The potential of AI use in clinical development and drug trials
Bringing a drug from initial identification through regulatory approval and onto the market requires substantial resources, including time, money, and coordination. The process, from screening potential compounds to candidate nomination, through human clinical trials until receiving regulatory approval, can take an average of 12 to 15 years1.
AI in drug discovery and early-stage development
The drug development process begins with basic research, searching for the right target for a particular medical problem. AI in trials is improving early drug discovery by accelerating compound screening and lead candidate identification. Historically, identifying a lead compound required laboratory screening of thousands of chemical structures2. Followed by a series of additional tests to tweak the lead compound to get it ready for clinical testing, including optimizing the drug properties for how the drug may interact inside the body and with its target.
After these steps, the lead candidate proceeds to clinical trials to determine if there is a safe and effective dose for patients. The clinical trial process itself can take years and is quite expensive. Even at this point, there can be a high rate of failure. This means the drug might not be safe at the desired dose or lacks efficacy for its intended medical condition3. In cancer drug development, it is estimated that the research and development costs to bring a successful drug to market runs between $0.9 to $2.1 billion4. Thus, finding more efficient ways to bring new therapies to patients is an ongoing endeavor. This is where artificial intelligence (AI) can help across the drug development continuum.
AI can facilitate drug development through several means, including machine learning (ML) and natural language processing (NLP). ML is a subfield of AI “that allows computers to learn without explicitly being programmed”5 like analyzing data or categorizing images. NLP is an AI that “enables computers and digital devices to recognize, understand, and generate text and speech”6.
Before human testing: AI in compound screening and optimization
1. AI in compound screening for lead candidate identification
AI can speed up the process of discovering new medicines by analyzing numerous chemical structures to identify the most promising candidates, enabling researchers to begin testing potential drugs sooner. Programs like AlphaFold enhance clinical trial intelligence by predicting protein’s three-dimensional structure based on its amino acid sequence. Improving protein structure prediction allows for better drug designs targeting that protein, reducing the number of chemical structures needed for laboratory screening, thereby increasing efficiency and reducing costs.
2. In silico experiments for compound optimization
AI can simulate how a lead compound and its modifications might interact in the human body. These simulations can significantly reduce timelines by identifying necessary structural modifications before confirmatory laboratory studies7.
Once the optimized lead candidate is ready for clinical testing in human trials, AI use in clinical development enhances multiple aspects, including trial design, patient recruitment, and treatment personalization.
3. AI use in clinical development and trials
AI can enhance clinical trial design by analyzing extensive past data and real-world evidence (RWE). This analysis can alert researchers to potential risks and pitfalls in patient enrollment and eligibility criteria, allowing for further study optimization7. In certain situations, this may also allow for a synthetic control arm in the study. Having a synthetic control arm can eliminate the need to enroll patients on the standard of care treatment arm of the study by using RWE from other sources. This has the possibility to cut a clinical trial’s cost by up to 50% (as half the original number of patients would need to be enrolled) and it could speed up approval by up to 1.5 years8. It should be noted that synthetic control arms are likely going to be reserved for situations where there is an established standard of care treatment and a fairly predictable course of disease.
4. Finding potentially eligible patients
AI can also assist in identifying suitable patients for clinical trials which is an involved and challenging task. Recruitment for clinical trials is hard and complex. Many AI drugs in clinical trials utilize AI-driven algorithms to analyze medical records to find potential participants7. Once patients are identified, deemed eligible, and begin participating in a study, retaining and keeping the patients engaged and adhering to the study requirements is also important. If too many patients drop out or exit the study early, the reliability of the study can be compromised. AI tools are also being deployed to help keep enrolled patients on track with their study participation requirements7.
5. Treatment personalization
AI can use data about a patient’s genes, biomarkers, and medical history to predict the most effective treatments. To take it further, AI can also predict how a patient might respond to treatment9 or alert their doctors about a potential side effect that may require alteration of the treatment10.
6. Future possibilities of AI in clinical trials
AI is poised to play a larger role in how AI is being used to accelerate clinical trials. Additionally, AI could help expedite regulatory submissions and review to further cut down on the timelines to regulatory approval of new safe and effective drugs for patients.
Case study: AI in immunotherapy Clinical Trials
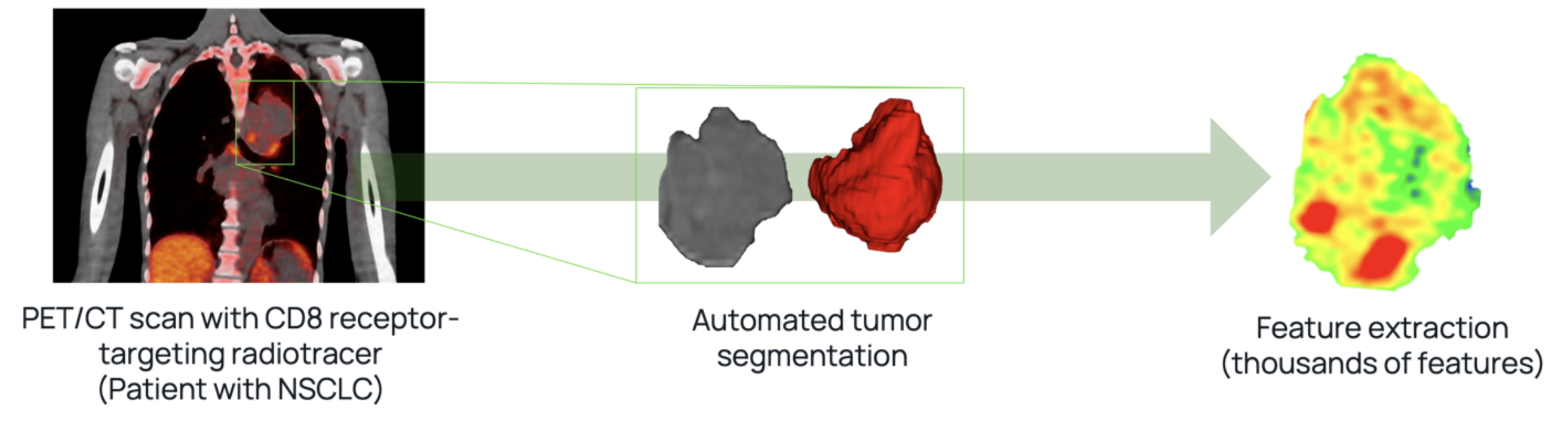
AI is reshaping clinical trial intelligence in immunotherapy research. In the therapeutic areas of melanoma, renal cell carcinoma, non-small cell lung cancer, head and neck squamous cell carcinoma, and others immunotherapy has shown remarkable promise. However, accurately predicting treatment efficacy remains a challenge. Novel radiotracers targeting the CD8 receptor on human T cells for quantitative, non-invasive PET imaging are under development. Quibim is creating an imaging-based algorithm to interpret PET/CT exams, aiming to quantitatively assess the efficacy of an 89Zr-labeled minibody in targeting the CD8 receptor in the tumor microenvironment. This technology enhances AI use in clinical development by providing precise, non-invasive methods to evaluate immunotherapy effectiveness. The resulting data provides valuable insights into the minibody’s biodistribution and targeting specificity, informing future clinical development and potential therapeutic applications.
AI is transforming clinical trials and artificial intelligence integration in drug development. From AI drugs in clinical trials to clinical trial intelligence, AI is reducing costs, improving efficiency, and personalizing treatment strategies. While challenges remain, the role of AI use in clinical development will continue to expand, shaping the future of medicine and accelerating access to life-saving treatments.
References
- Hughes JP, Rees S, Kalindjian SB, Philpott KL. Principles of early drug discovery. Br J Pharmacol. 2011;162(6):1239-1249. doi:10.1111/j.1476-5381.2010.01127.x
- Hefti FF. Requirements for a lead compound to become a clinical candidate. BMC Neurosci. 2008;9 Suppl 3(Suppl 3):S7. Published 2008 Dec 10. doi:10.1186/1471-2202-9-S3-S7
- Jain R, Subramanian J, Rathore AS. A review of therapeutic failures in late-stage clinical trials. Expert Opin Pharmacother. 2023;24(3):389-399. doi:10.1080/14656566.2022.2161366
- Schlander M, Hernandez-Villafuerte K, Cheng CY, Mestre-Ferrandiz J, Baumann M. How Much Does It Cost to Research and Develop a New Drug? A Systematic Review and Assessment. Pharmacoeconomics. 2021;39(11):1243-1269. doi:10.1007/s40273-021-01065-y
- https://mitsloan.mit.edu/ideas-made-to-matter/machine-learning-explained
- https://www.ibm.com/topics/natural-language-processing
- Hutson M. How AI is being used to accelerate clinical trials.Nature. 2024;627(8003):S2-S5. doi:10.1038/d41586-024-00753-x
- https://www.statnews.com/2019/02/05/synthetic-control-arms-clinical-trials/#:~:text=By%20reducing%20or%20eliminating%20the,speed%20lifesaving%20therapies%20to%20market
- Zhang B, Shi H, Wang H. Machine Learning and AI in Cancer Prognosis, Prediction, and Treatment Selection: A Critical Approach.J Multidiscip Healthc. 2023;16:1779-1791. Published 2023 Jun 26. doi:10.2147/JMDH.S410301
- Lippenszky L, Mittendorf KF, Kiss Z, et al. Prediction of Effectiveness and Toxicities of Immune Checkpoint Inhibitors Using Real-World Patient Data.JCO Clin Cancer Inform. 2024;8:e2300207. doi:10.1200/CCI.23.00207

