
The range of therapeutic options for cancer treatment along the patient journey is diverse, including surgery, radiation therapy, chemotherapy, hormone or targeted therapy, immunotherapy, or palliative care. These decisions can really alter patient-reported outcome measures, the effects on quality of life and independence, cancer recurrence, and short- and long-term side effects. Modeling these potential outcomes alongside mortality risk to provide personalized, optimized care may seem a moonshot in healthcare, but we have the opportunity to learn from other industries and domains of engineering to do this and convert it into a feasible tool for tomorrow’s Medicine.
What is digital twin imaging?
The digital twin concept emerged from NASA in the early 2000s and is now a staple of engineering, automotive, and aerospace industries. A digital twin is a virtual representation that serves as a real-time digital counterpart of a physical object or, in the case of medical imaging, the patient. For patient data, a digital twin approach can be used to anticipate the treatment responses of an individual patient as well as help in population-level studies to identify what healthy looks like 1.
The digital twin imaging approach also enables the real-time monitoring and modification of interventions depending on patient responses and the planning of surgical interventions through the development of physical monitoring of complex patients. This information will be invaluable to a precision medicine future, improving clinical decision making, drug development, and improving our fundamental knowledge about life-changing diseases.
The real value of a digital twin imaging will be in the improved diagnosis, monitoring, and personalized treatment for patients enabling clinicians to handle large amounts of patient data. This can allow more effective care and simulation of treatment paradigms. However, this depends on the quality and consistent reporting of data. We are a short while away from the day-to-day use of digital twins in our healthcare systems, and there are several challenges to overcome.
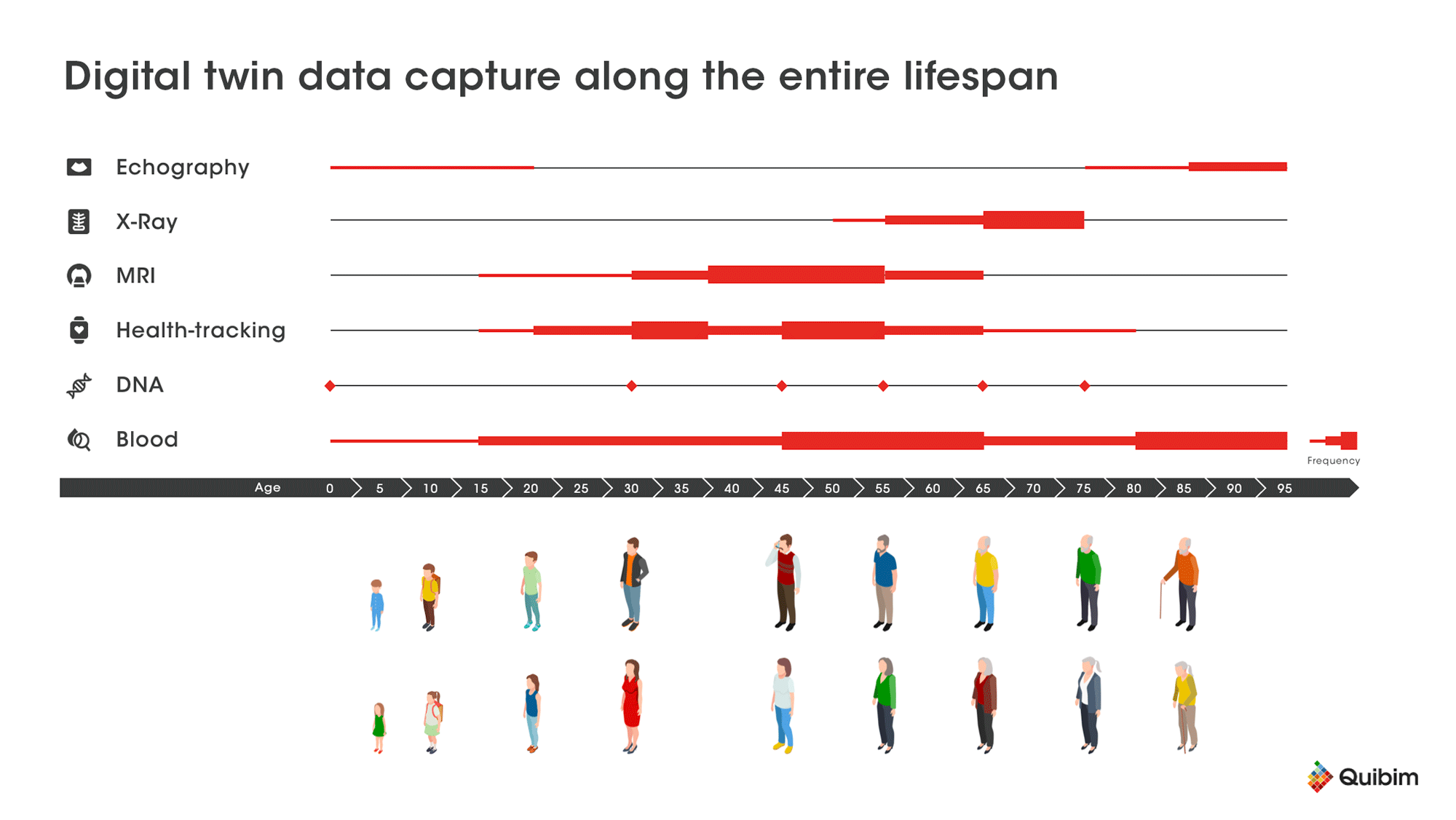
Digital twins imaging relies on the data quality and accuracy of the information put into the system. In addition, there are challenges around data management, algorithm design, Data Security and privacy, and ethical considerations that need to be overcome 2
For the best algorithm design, the accurate recording of data from existing patients and large population studies such as the UK biobank will provide the foundations for this and allow us to understand as a clinical and scientific community the reflective importance of clinical outcomes, multi-omics, imaging biomarkers, patient-related data, patient circumstances such as socioeconomic backgrounds and medical procedures outcomes that need to be understood to create the most effective digital twins.
In the context of medical imaging, this may include providing this information from many sources, such as combining Magnetic Resonance Imaging (MRI), Positron Emission Tomography (PET), Ultrasound, and Computer Tomography (CT). However, despite the significant progress in AI-based segmentation, the huge amount of time required for the manual or even semi-automatic segmentation of some difficult to delineate organs and lesions (i.e. pancreas cancer, head & neck cancer) for the extraction of imaging biomarkers from these modalities represents a clear barrier to the use of a digital twin approach.
Role of radiomics in a digital twin approach
Radiomics is a method that extracts a large number of features from medical images to allow clinicians to enhance the existing data available. Radiomics allows artificial intelligence to analyze cross-modality images providing detailed, accurate information on shape, pixel intensity, texture, and also filtering of images to help aid the diagnosis decision process and estimate prognostic factors 3.
It is this automation accuracy that means radiomics is the ideal partner to the digital twin approach for the future of healthcare, providing the automated analysis of changes in tumor size, shape, and texture alongside changes in body composition such as loss of muscle and fat which may indicate changes in mortality risk through development of cachexia.
Whilst radiomics may provide the backbone for the speeding up on the segmentation and processing of medical images, especially in imaging biomarkers which are currently being trialed however, how these imaging biomarkers might alter with clinical interventions is still work in progress.
More real-world evidence (RWE) studies and meta-analysis on existing literature are needed to be able to generate actionable links between radiomics and the clinical endpoints, allowing the creation of imaging biomarker panels that can be used routinely in the management of patients with cancer within the healthcare system.
How will digital twin imaging support the raiolodist of the future?
Digital twin imaging technology will be a key enabler for radiologists of the future, offering a dynamic virtual representation that will complement physical systems in real time. From the development of digital twins will come advances in clinical practice by enabling patient-specific predictions, such as prevention, disease diagnosis, and treatment customization. Radiology’s ability to generate huge amounts of data from various modalities, such as CT and MRI, will also be key in the creation of these digital twins.
Radiologists will be able to use digital twins imaging to make more accurate predictions, improve treatment planning, and assess therapeutic response on an individualized basis. In addition, digital twins imaging will not only optimize clinical and hospital throughput, but will also be used in research, development of new medical devices, and training. Although there are technical and ethical challenges, such as data integration and the creation of an appropriate regulatory framework, the potential benefits of this technology-including improved diagnostic and treatment accuracy without intervening on actual patients-make it a powerful tool for the future of radiology.
Conclusion
The importance of digital twin imaging technologies in the future of personalized medicine is on the brink of becoming a reality; this will offer improvements in patient clinical outcomes as well as more economical and efficient healthcare industry. Technological innovations such as Radiomics will provide the foundation for this in offering an accurate, cost-effective, timely analysis of multi-modal imaging data.
Quibim projects on the digital twin concept

References
- Digital twins in health care: Ethical Implications of an emerging engineering paradigm (2018). Bruynseels K; Santoni de Sio F, van den Hoven J. Frontiers in Genetics 9:31.
- Digital Twins for Multiple Sclerosis (2021). Voigt I, Inojosa H, Dillenseger A et al., Front Immunol. July 2021.
- Radiomics in medical imaging- “how-to” guide and critical reflection (2020). van Timmeren JE, Cester D, Tanadini-Lang S, Alkadhi H,Baessler B. Insights into Imaging 11;91.
- Imaging biomarker roadmap for cancer studies (2017). O’Connor JPB, Aboagye EO, Adams JE et al., Nature Reviews Clinical Oncology 14,169-186.

